|
van der Wall’s forces are weaker hence the distance between the atoms is larger. Generally, atomic radii of inert gases are expressed in terms of van der Walls’ radius. the closest distance between two hydrogen atoms without forming the bond is 240 pm. Hence van der Walls’ radius of sodium = 240 / 2 = 120 pm. Van der Walls’ radius is defined as one half of the distance between the nuclei of the two atoms of the same substance at their closest approach. This property can be used to find the internuclear distance between two molecules forming a single covalent bond among themselves. Hence crystal radius of oxygen = 132 / 2 = 66 pm. the distance between two oxygen atoms in molecular oxygen is 132 pm. Hence crystal radius of sodium = 372 / 2 = 186 pm.Ĭovalent radius is defined as one half of the distance between the centres of the two similar nuclei of two similar atoms bonded together by a single covalent bond. For e.g. the distance between two sodium atoms in a sodium crystal is 372 pm. It is defined as one half of the distance between the centres of nuclei of two adjacent atoms in a metallic crystal. Other Terms Related to Atomic Radius: Crystal Radius: Hence the definition given in above point is arbitrary. The electron density in an atom is greatly influenced by the presence of other atoms around the bonding atom and the nature (type) of bonding with neighbouring atoms, Depending upon this the terms like crystal radius, covalent radius, van der Walls’ radius, tetrahedral radius, etc. The atomic radius (atomic size) may be regarded as the distance from the centre of the atom to the outermost (valence) shell of electrons. 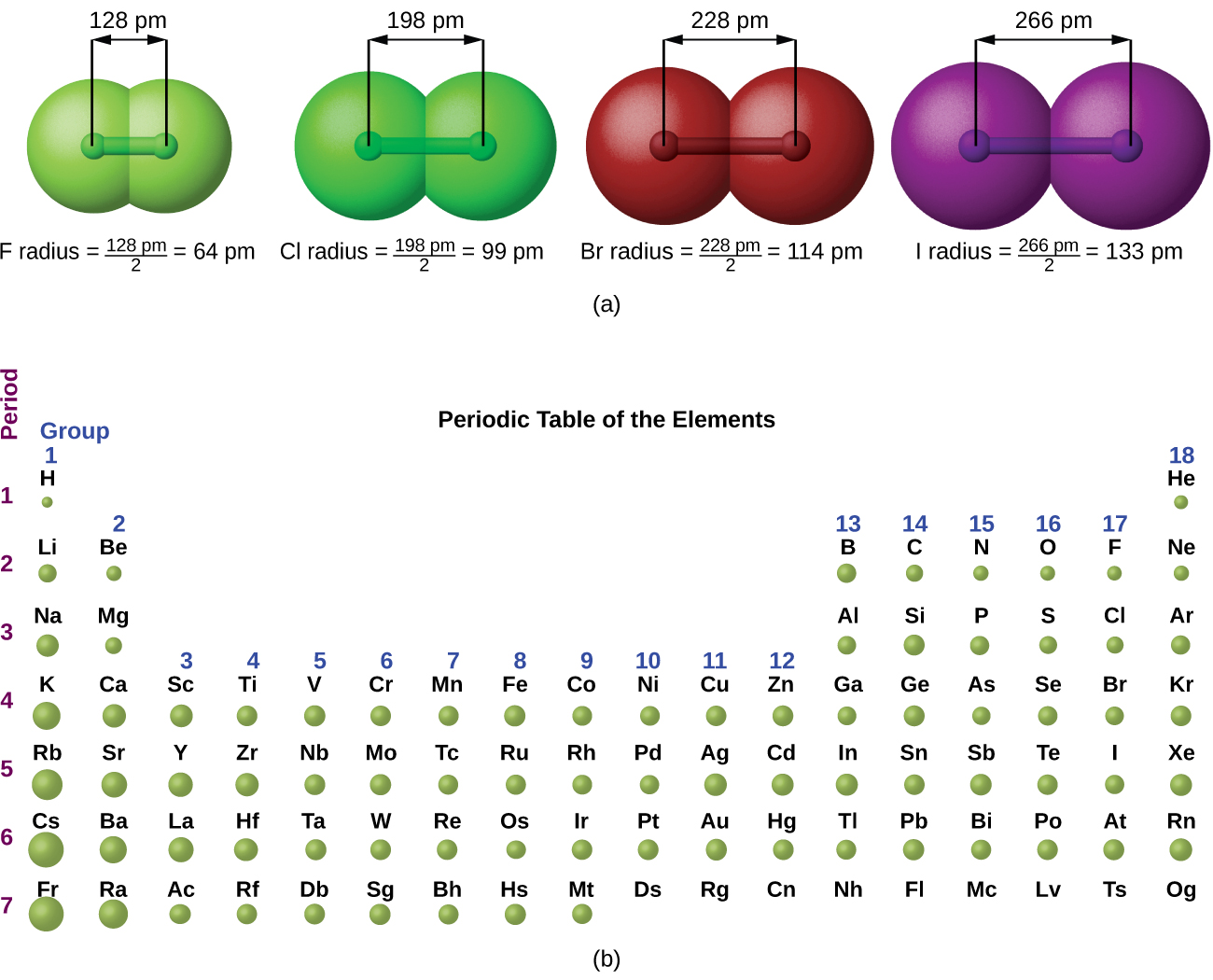
One practical approach of finding the size of an atom of a non-metallic element is to measure the distance between two atoms when they are bound together by a single bond in a covalent molecule and from this value, the “Covalent Radius” of the element can be calculated. 
Secondly, since the electron cloud surrounding the atom does not have a sharp boundary, the determination of the atomic size cannot be precise. The size of an atom is very small (120 pm).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
